00120
Neutron and Synchrotron Diffraction Studies on Structural Phase Transitions in 1:3 Ordered Perovskites Sr 4-xBaxNaSb3O12
School of Chemistry, The University of Sydney
○Qingdi Zhou Brendan J Kennedy
As part of our ongoing studies of the structures and phase transitions in perovskites oxides xx members of the series of 1:3 ordered perovskites of the type Sr4-xBaxNaSb3O12 have been synthesized and their structures determined using synchrotron X-ray and neutron powder diffraction techniques. At room temperature Ba4NaSb3O12 has a cubic structure, where the Na and Sb cations are ordered but there is no tilting of the octahedra. As the average size of the A-site cation decreases tilting of the octahedra is introduced and ultimately Sr4NaSb3O12 presents a monoclinic structure in C2/c. The powder neutron diffraction studies show that the two monoclinic phases of C2/c and P21/n co-exist at room temperature for samples with x between 1 and 2.5.
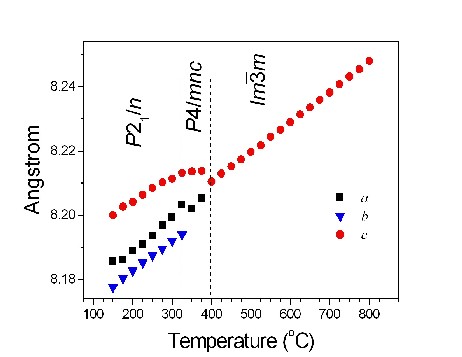
Variable temperature synchrotron powder diffraction studies demonstrate that the C2/c rapidly transforms to P21/n upon heating. A transition from monoclinic P21/n to tetragonal P4/mnc occurs at ~350 C for Sr2.5Ba1.5NaSb3O12, and again, a continuous transition to the cubic structure near 400 C (Fig 1). These experimental results are consistent with a group theoretical analysis of the phase transition in 1:3 ordered perovskites[1].
References
Howard, C.J. and Stokes, H.T. (2004) Acta Cryst. B60, 674-684.